EHRA-PATHS
EHRA-PATHS: Addressing multimorbidity in elderly atrial fibrillation patients through interdisciplinary, patient-centred, systematic care pathways.
EHRA-PATHS
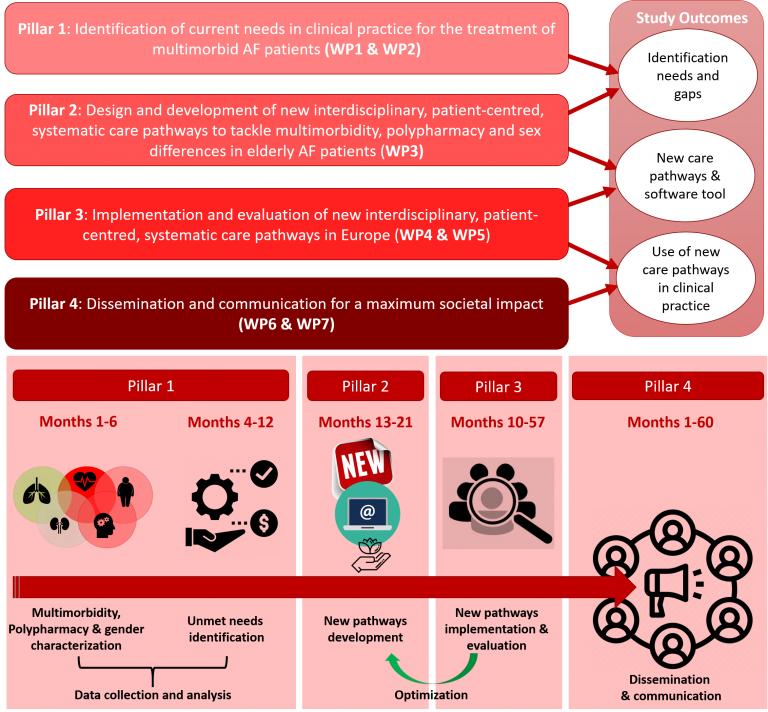
The EHRA-PATHS project aims to transform and improve the clinical practice in the field of atrial fibrillation to holistic, inclusive and personalised treatment strategies. It will change the single-disease focused approach to a multi-factorial approach considering multimorbidity, polypharmacy, and sex.
Approach: The EHRA-PATHS approach is built on 4 pillars:
1. Identification of current needs in clinical practice for the treatment of multimorbid, elderly AF patients (WP1 & WP2)
2. Design and development of new interdisciplinary, patient-centred, systematic care pathways to tackle multimorbidity, polypharmacy and sex differences in elderly AF patients (WP3)
3. Implementation for systematic and uniform adoption and evaluation of these new care pathways in Europe (WP4 & WP5)
4. Dissemination and communication for a maximum societal impact (WP6 & WP7)
Work package 5 (WP5) – UMCG
Objective: To implement and evaluate new care pathways to systematically assess and initiate treatment for multimorbidity in elderly AF patients.
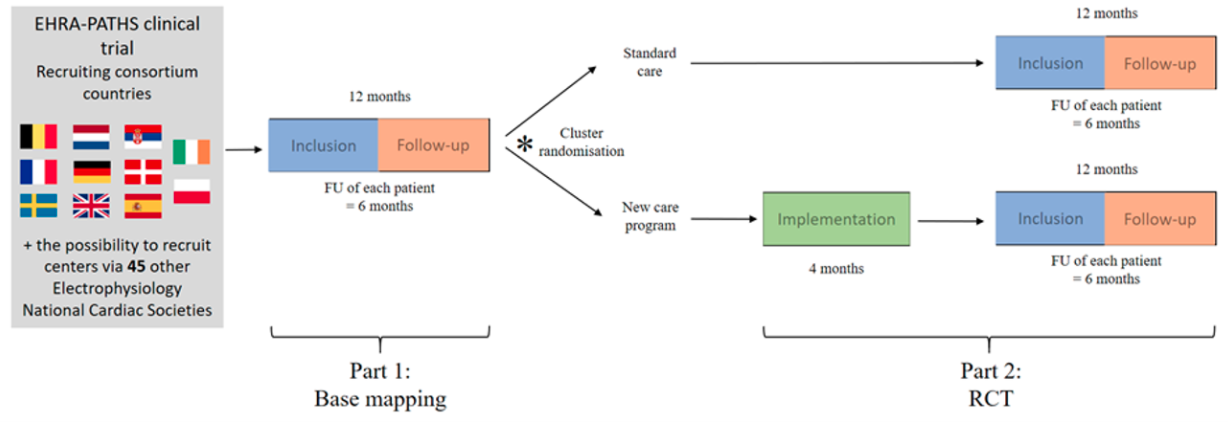
Study design: A prospective, multicentre, international study consisting of two parts. Part 1 will consist of a prospective observational trial of each site’s standard care pathways, which will serve as “base mapping” of the current management of comorbidities in elderly patients newly diagnosed with atrial fibrillation. Part 2 will be a prospective, cluster-randomised controlled study at the same centres to investigate if the newly developed care program leads to a better evaluation of comorbidities and adequate treatment initiation in new AF patients.
Study population: newly diagnosed AF patients ≥65 years of age
Primary endpoint part 1: the number of comorbidities that are formally evaluated and for which tereatment is adequately initiated within 6 months of a new AF diagnosis.
Primary endpoint part 2: the implementation of a risk management program resulting in all comorbidities that are formally evaluated and for which an adequate treatment is initiated within 6 months of a new AF diagnosis.
Funding
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 945260
People involved
Awards and funding