Red – light – sensitive BODIPY photoprotecting groups for amines and their biological application in controlling heart rhythm
Publication: Heart rate control with light-sensitive drugs
Heart failure patients usually take medication to manage their symptoms. These drugs are taken orally, which results in high initial concentration in the blood before they are cleared and concentration gradually drop. This way, drug concentrations are only shortly within the desired ranges for an optimal effect. Side effects can occur when concentrations become too high and drug efficacy is suboptimal at low concentrations.
Novel chemical approaches are being investigated to reduce adverse effects of drugs while retaining its desired function. The field of photopharmacology focusses on the development of photo-activated drugs. These drugs can, for example, be encapsulated in A protective chemical “cage” that is removed upon exposure to light. The uncaged drugs are biologically active and can exert their function. Consequently, administered dosages of a chemically protected do not reflect active drug concentrations in the circulation. The desired active concentrations can be titrated by illuminating the protected drugs with high precision in space and time.
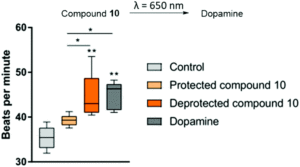
These approaches were only compatible with UV light, which reduced their biocompatibility. The group of Ben Feringa and Wiktor Szymanski has been working on making this approach more clinically applicable by red-shifting the protective cage (i.e. making it compatible with red light instead of harmful UV light). In a multidisciplinary collaboration, Martijn Hoes and Peter van der Meer were able to employ human stem cell-derived cardiomyocytes and confirm that exposure to red light indeed activated a caged form of dopamine. The beating frequency of these cardiomyocytes increased after exposure to red light. The results have been published by the Royal Society of Chemistry in Chemical Communications.